UNC School of Medicine scientists led by Eva Anton, PhD, found that defective primary cilia can modulate connectivity between neurons during brain development and cause rare neurodevelopmental conditions called Joubert-syndrome related disorders.

UNC School of Medicine scientists led by Eva Anton, PhD, found that defective primary cilia can modulate connectivity between neurons during brain development and cause rare neurodevelopmental conditions called Joubert-syndrome related disorders.

Media contact: Mark Derewicz, 984-974-1915
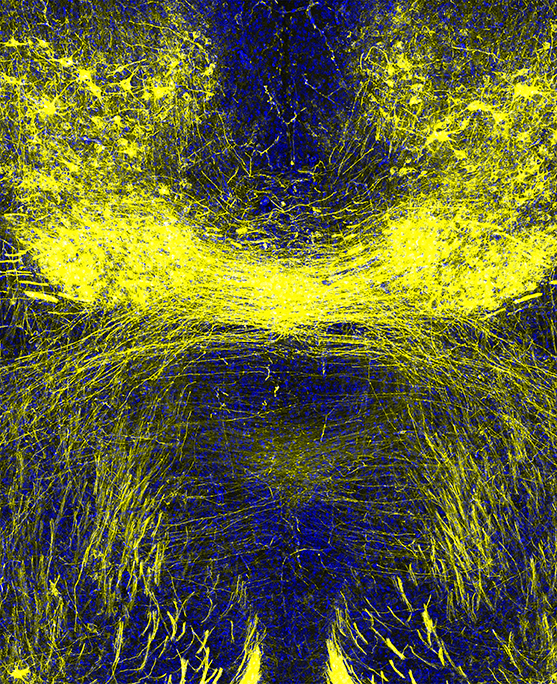
CHAPEL HILL, NC – December 16, 2019 – Axons are the long thread-like extensions of neurons that send electrical signals to other brain cells. Thanks to axonal connectivity, our brains and bodies can do all necessary tasks. Even before we’re born, we need axons to grow in tracts throughout gray matter and connect properly as our brains develop. UNC School of Medicine researchers have now found a key reason why connectivity goes awry and leads to rare but debilitating neurodevelopmental conditions.
Published in the journal Developmental Cell, researchers led by Eva Anton, PhD, professor of cell biology and physiology at UNC-Chapel Hill, show how two gene mutations alter the function of neuronal cilia – antennae-like protuberances found on many cell types. The resulting dysfunctional cilia affect axonal connectivity and leads to rare Joubert syndrome-related disorders (JSRD).
“Our experiments demonstrate that ciliary signaling facilitates appropriate patterns of axon tract development and connectivity,” said Anton, who is a member of the UNC Neuroscience Center. “Disrupting ciliary signaling can lead to axonal tract malformations in JSRD.”
Although cilia are found on most cell types, their significance in brain development, has been largely underappreciated, until recently.
Scientists now know that cilia sense the environment around them, and dysfunctional cilia mess up axonal growth and connectivity during fetal development. Babies born with dysfunctional cilia and associated irregular axonal growth and connectivity can develop JSRD. Molar tooth sign, a characteristic defect of axonal projections detectable in brain MRI images, is often used to diagnose JSRD. People with the condition experience developmental delays, intellectual disabilities, abnormal respiratory rhythms, trouble controlling their body movements, and other serious health issues. But how this happens has not been clear.
Using neuron-specific mouse genetic models of two genes called Arl13b and Inpp5 and related human mutations from JSRD patients, as well as chemo-genetic and opto-genetic manipulation of primary cilia signaling, Anton and colleagues investigated how cilia become dysfunctional and affect axonal connectivity during brain development.
In mice, they found that deletion of Arl13b or Inpp5e impairs the ability of the primary cilium to function as a signaling hub, thus allowing them to examine how cilia-driven signaling regulates axon growth and connectivity in normal and JSRD brains. Anton and colleagues went on to delineate ciliary-driven changes in cell signaling, particularly the ones mediated through major signaling proteins PI3K AKT, and AC3 effectively modulate axonal behavior.
Before this research, the significance of primary cilia in the emergence of brain connectivity were undefined. Nor did the research community understand exactly how cilia dysregulation led to axonal tract defects in Joubert syndrome-related disorders.
“By shedding light on the significance of primary cilia in the emergence of brain connectivity, this research helps us understand how cilia dysregulation led to axonal tract defects in Joubert syndrome-related disorders,” Anton said. “Our studies indicate precise manipulation of ciliary signaling in the future may be tested and utilized to alleviate neuronal connectivity defects in ciliopathies, such as JSRD.”
This research was funded by the National Institutes of Health and the Natural Sciences and Engineering Research Council of Canada.
Other authors are Jiami Guo, James Otis, Sarah Suciu, Christy Catalano, Lei Xing, Sandii Constable, Dagmar Wachten, Stephanie Gupton, Janice Lee, Amelia Lee, Katherine Blackley, Travis Ptacek, Jeremy Simon, Stephanie Schurmans, Garret Stuber, and Tamara Caspary.